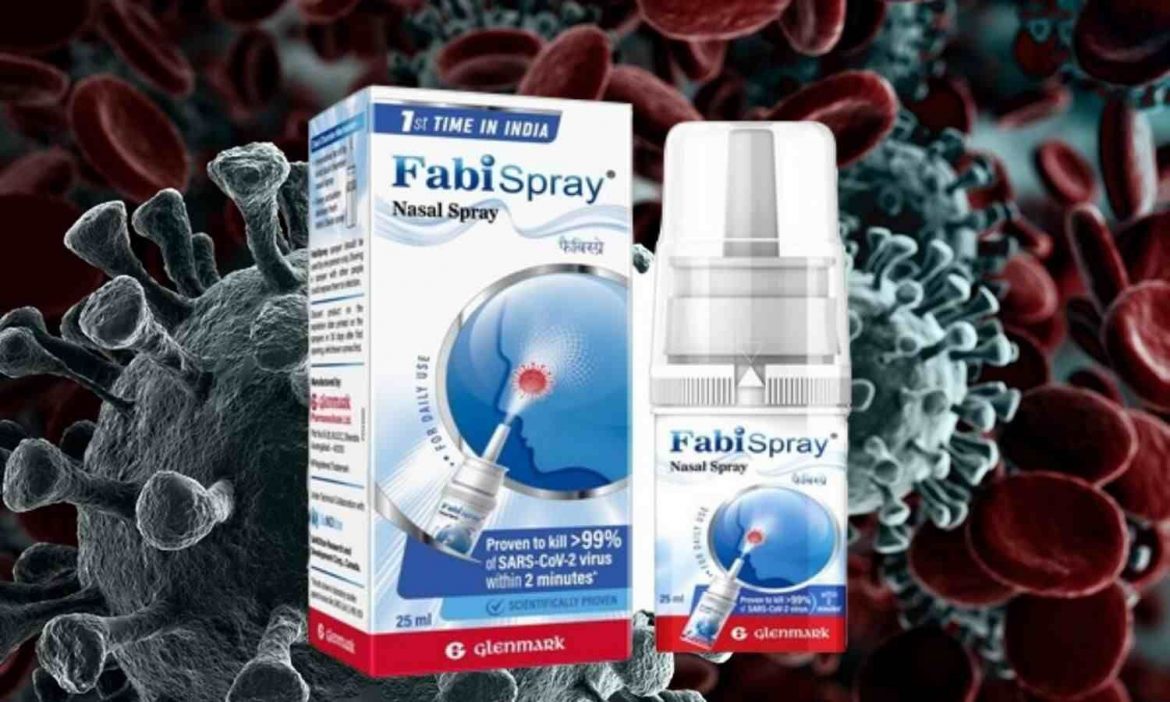
As part of an accelerated approval process, the Mumbai-based pharmaceutical company gained manufacturing-marketing authorisation for Nitric Oxide Nasal Spray from India’s drug regulator, the Drugs Controller General of India (DCGI).
Glenmark Pharmaceuticals launched a nasal spray in India on Wednesday to treat adult patients with coronavirus illness (Covid-19). In collaboration with Canadian pharmaceutical business SaNOtize Research & Development Corp, FabiSpray, a Nitric Oxide Nasal Spray, has been released.
The Mumbai-based pharmaceutical company received manufacturing-marketing approval from India’s drug regulator, Drugs Controller General of India (DCGI) for Nitric Oxide Nasal Spray as part of an accelerated approval process. “Phase 3 trial in India met the key endpoints and demonstrated reduction of viral load of 94 per cent in 24 hours and 99 per cent in 48 hours. Nitric Oxide Nasal Spray (NONS) was safe and well-tolerated in Covid-19 patients. Glenmark to market NONS under the brand name FabiSpray,” reads the official statement.
FabiSpray is designed to kill the Covid-19 virus in the upper airways. As per the company’s claims, the Nitric Oxide Nasal is sprayed over nasal mucosa which then acts as a physical and chemical barrier against the virus. It will prevent the virus from incubating and spreading to the lungs, the statement reads.
“This reaffirms our commitment of providing yet another safe and effective antiviral treatment for Covid-19, and we are confident that it will offer patients a much needed and timely therapy option,” Robert Crockart, chief commercial officer of Glenmark Pharmaceuticals Ltd, said in the statement.
The company had entered into an exclusive long term strategic partnership with Canada’s SaNOtize to manufacture and market the nasal spray in July 2021. The spray will also be distributed to other Asian markets including Singapore, Malaysia, Hong Kong, Taiwan, Nepal, Brunei, Cambodia, Laos, Myanmar, Sri Lanka, Timor-Leste and Vietnam.
Meanwhile, the country on Wednesday logged 71,365 fresh cases in the last 24 hours, pushing the overall tally to reach 4,24,10,976. The death toll surged to 505,279.
Meanwhile, the number of active cases has fallen below the 9,00,000 mark as the caseload fell at 8,92,828, the data also showed.