In a significant breakthrough for cancer treatment in India, the country’s drug regulator, the Central Drugs Standard Control Organisation (CDSCO), has approved Qartemi, a second-generation Chimeric Antigen Receptor T-cell (CAR-T) therapy for blood cancer. Developed by Immuneel Therapeutics, a Bengaluru-based biotech startup, Qartemi is being hailed as a “living drug” due to its innovative use of genetically modified T-cells to treat blood cancers like B-cell Non-Hodgkin Lymphoma (B-NHL). This marks a monumental shift in the treatment of blood cancers, offering hope to thousands of patients who have exhausted all other treatment options.
What is CAR-T Therapy?
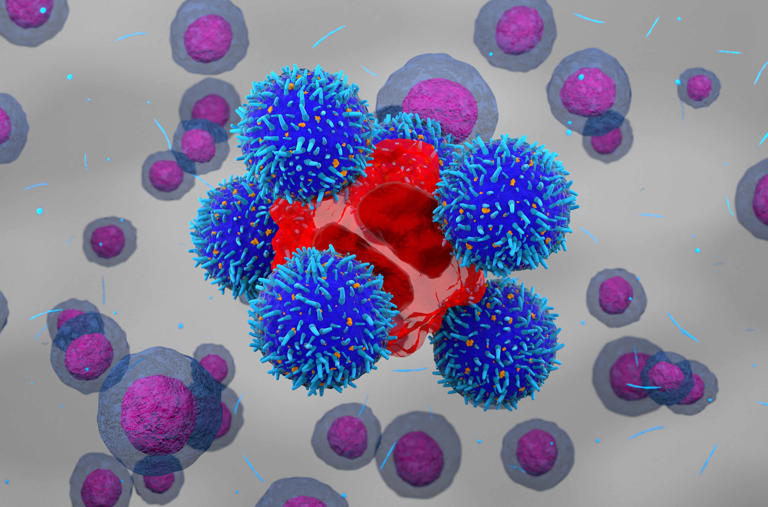
CAR-T cell therapy is a revolutionary form of immunotherapy, which utilizes the patient’s own immune cells to fight cancer. Unlike traditional chemical drugs, CAR-T cell therapy involves extracting T-cells (a type of white blood cell) from a patient’s blood, genetically modifying them to better identify and attack cancer cells, and then infusing them back into the patient’s bloodstream. This “living drug” provides a long-lasting immune response against cancer cells, which can be particularly effective in cases where conventional therapies, such as chemotherapy, have failed.
Qartemi, developed by Immuneel Therapeutics, is the second CAR-T therapy to be approved in India after NexCAR19, developed by ImmunoAct. The drug targets adult patients suffering from relapsed or refractory B-NHL, a cancer that affects the immune system. This approval brings significant hope to patients with advanced stages of blood cancer who have limited treatment options.
Qartemi: A Game-Changer for Blood Cancer Treatment
India has been grappling with a rising burden of blood cancers, with around 120,000 new cases and more than 70,000 deaths annually from leukemia, lymphoma, and multiple myeloma. Many of these patients do not respond to traditional treatments like chemotherapy, making it crucial to develop innovative therapies like Qartemi.
The launch of Qartemi is a significant milestone in cancer care in India, as it offers a cutting-edge, personalized treatment option for patients who have not responded to other therapies. This CAR-T therapy has been developed to be cost-effective compared to similar global alternatives, making it accessible to a larger number of patients. Priced between Rs 35 lakh to Rs 50 lakh, Qartemi provides a more affordable option than its international counterparts, which can cost upwards of a crore rupees.
Amit Mookim, CEO of Immuneel Therapeutics, expressed pride in developing Qartemi indigenously, ensuring that it meets global standards of safety and efficacy while being accessible to Indian patients. The company aims to transform the landscape of blood cancer treatment in India by offering an advanced, localized solution to the rising cancer burden.
Clinical Trials and Results
The clinical trials for Qartemi, known as the IMAGINE trial, were conducted at some of India’s leading medical institutions, including Narayana Health in Bengaluru, Apollo Cancer Hospital in Chennai, and PGIMER in Chandigarh. These trials focused on patients with relapsed or refractory B-NHL, who had limited options due to their disease not responding to conventional drugs.
The results of the IMAGINE trial were highly promising, with an 83.3% overall response rate, demonstrating that Qartemi was both safe and effective. The drug’s efficacy was found to be comparable to other CAR-T cell therapies that have been approved by the US Food and Drug Administration (FDA), highlighting its potential to provide transformative care for blood cancer patients.
The trials were supported by the Department of Biotechnology’s Agency (BIRAC) through its Biotechnology Industry Partnership Programme (BIPP) scheme, ensuring that the therapy met rigorous standards of safety and effectiveness. The approval of Qartemi represents a significant leap in India’s cancer care capabilities, showcasing the country’s potential to develop and deliver world-class medical innovations.
The Role of Immuneel Therapeutics
Immuneel Therapeutics, the biotech startup behind Qartemi, has been instrumental in developing this groundbreaking therapy. Founded with a mission to revolutionize cancer treatment, Immuneel is led by experts in the fields of immunotherapy, biotechnology, and oncology. Backed by prominent investors such as Biocon founder Kiran Mazumdar Shaw and oncologist Dr. Siddhartha Mukherjee, the company has made substantial strides in the field of gene and cell therapies.
The company’s focus on CAR-T cell therapies is aligned with the growing demand for innovative cancer treatments that can address the limitations of conventional therapies. Immuneel’s commitment to making Qartemi an accessible and affordable treatment option for Indian patients reflects its vision of making cutting-edge therapies available to the broader population.
Availability and Partnerships
Qartemi is now available locally for patients with relapsed or refractory B-NHL. Immuneel Therapeutics has partnered with several prestigious medical institutions across India to provide the drug to eligible patients. Hospitals such as Narayana Health, Apollo Hospitals, CMC Vellore, Manipal Hospitals, and PGIMER Chandigarh, among others, have been included in the network to administer the therapy to patients.
The therapy’s availability in these hospitals will enable a large number of patients to benefit from this life-changing treatment. In addition to making Qartemi available to Indian patients, Immuneel Therapeutics has also partnered with renowned global institutions, such as Hospital Clínic de Barcelona, to license the therapy and ensure that it meets international standards.
Global Impact and Future of CAR-T Therapy in India
Qartemi is poised to make a significant impact not only in India but also in the global medical community. As the second CAR-T cell therapy approved in India, it represents the country’s growing role in the global biotechnology and pharmaceutical industry. By developing and manufacturing cutting-edge therapies like Qartemi locally, India is positioning itself as a hub for innovation in cancer care and biotechnology.
Looking ahead, Immuneel Therapeutics plans to expand its research and development efforts to explore other potential applications of CAR-T cell therapy for various types of cancers. The success of Qartemi is expected to pave the way for the development of additional immunotherapies that could revolutionize the treatment of other cancers, providing new hope for patients who currently have few options.
In the coming years, India’s role as a leader in the biotechnology and medical innovation sectors is set to grow. With increasing collaboration between local biotech companies, government agencies, and global institutions, India is well-positioned to become a global leader in cell and gene therapies.
The approval of Qartemi in India is a major breakthrough for blood cancer treatment, offering hope to thousands of patients battling B-cell Non-Hodgkin Lymphoma and other blood cancers. As the second CAR-T cell therapy approved in the country, Qartemi demonstrates India’s growing capabilities in biotechnology and immunotherapy. With a focus on affordability, accessibility, and safety, Qartemi represents a new era of cancer care in India, ensuring that cutting-edge treatments are available to those who need them the most.
As India continues to make strides in the field of medical innovation, the success of therapies like Qartemi could pave the way for even more advancements in the treatment of cancer and other diseases, solidifying the country’s position as a global leader in healthcare and biotechnology.